NEOV LASER 1470
The neoV1470 is the workhorse of our surgical platform, representing the large majority of our installed base. The neoV1470 is a superb microsurgical instrument for endovenous laser ablation of veins, as well as for various key indications in proctology. The peak of water absorption at 1470nm enables precise cutting with good coagulation of soft tissue. As a result of this unique tissue absorption, procedures can be completed at lower energy dosages leading to less thermal trauma and better healing.

ABOUT US
neoLaser is a world leader in design, engineering and manufacturing of medical and surgical laser systems. neoLaser offers top-notch quality and world-class design, providing superb performance and functionality, flexibility and modularity, all at an affordable cost.
neoLaser’s neoV laser platform supports a variety of high-volume applications in Endovascular and Proctology surgery as well as spine and aesthetic procedures.
Founded by seasoned veterans of the medical laser industry, neoLaser brings together the highest engineering, marketing and design knowledge. The company is ISO13485:2016 certified, including MDSAP certification, and holds registrations in many geographical areas including CE, FDA, Health Canada, ANVISA Brazil, TGA Australia and more.
This website and its content as it relates to products and applications is supplied for EU customers. For non-EU based customers, please contact neoLaser or your local neoLaser representative to determine which products and applications are applicable per existing non-EU clearances.
WHY US ?
QUALITY
From the first day of operations, we have placed product quality as our number one priority. We believe this is the only viable long-term path to success and sustainability. Quality is our focus in product efficacy, in product safety, in customer service and support, and in any aspect of our company operations. neoLaser has established, maintained, and developed the most rigorous Quality System possible, leading to product registration in many key markets including USA (FDA), Europe (CE mark), Australia (TGA), Brazil (Anvisa), Canada (Health Canada), Israel (AMAR), Taiwan (TFDA), and many others. The company holds the ISO13485:2016 certification.
ACCEPTANCE
The neoV platform, launched in 2013, has achieved phenomenal growth and acceptance on a global scale. Today, it is the tool of choice in over 1,000 hospitals, surgical centers, and clinics, in over 40 countries, having been used in over 200,000 medical interventions to date with a growth rate of 35-40% every year. It has become a true sign of excellence in technology, product design, and clinical results.
EXPERIENCE & VALUES
neoLaser was founded, developed and built by a group of seasoned and experienced individuals, focused on surgical laser technology, and having decades of relevant industry knowledge. The neoLaser team has been responsible for multiple successful surgical laser product launches in a variety of geographies and in multiple surgical disciplines.
Our core values include integrity, humility, intellectual curiosity and rigor, combined with a constant and aggressive strive for excellence in all we do. As a young and agile company, we understand the needs of our distributors, physicians and patients, react very quickly, and are connected 24/7 to support our customer base, offering the best possible service. We are open to feedback and strive to dominate our industry by providing optimal clinical results through excellent, precise, stable, safe and effective product
Endovenous Laser Ablation - FIBERS
CORONA Infinite Ring Fiber
The CORONA Infinite Ring Fiber is the newest addition to the CORONA fibers family. The CORONA Infinite Ring Fiber is a groundbreaking development for endovenous laser treatment of the vena saphena magna and the vena saphena parva in patients with venous insufficiency. Providing radial emission from a continuous section of ~4mm length, it is the first ever fiber to enable optimal uniformity of energy deposition ALONG the vein wall. This leads to potential reduction in sticking, carbonization and excessive thermal damage; properties of special importance when working in critical areas which are sensitive to complications.
The fiber tip is fused to the fiber body for optimal safety, and as such, presents the latest fiber technology currently available on the market for endovenous laser treatments.

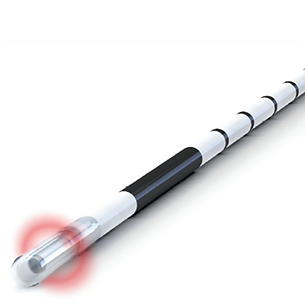
CORONA 360 Fused Fiber
The 360° emitting medical probe serves for the EVLA treatment of the vena saphena magna and the vena saphena parva in patients with venous insufficiency. With its 360° emission and optimal handling, the CORONA 360 is a superb, safe and effective product for medical applications in phlebology. The ink free laser marking eases positioning of the probe inside the vein. All fibers are provided with a fused tip for extra safety during the procedure. The fiber is available in two configurations – standard size compatible with 6FR introducers, and slim size compatible with 4FR introducers and micro-puncture kits.
PROCTOLOGY
CORONA Fistula Probe
The CORONA Fistula Probe is a special fiber with radial emission and optimized markings to allow treatment of both complex anal fistula procedures, as well as pilonidal sinus cases. The fiber includes a fully fused quartz tip for extra safety.


CORONA Hemorrhoid Probe
The CORONA Hemorrhoid Probe is a unique fiber optimized for treatment of grade 2-4 hemorrhoids. A combination of wide emission and sharp tip, allows easy insertion of the probe into the hemorrhoidal pile, and gentle application of energy to allow disruption of underlying vasculature.
